Kolekce 98 Atom Of Nitrogen Diagram
Kolekce 98 Atom Of Nitrogen Diagram. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …
Prezentováno Solved To Answer The Questions Interpret The Following Lewis Diagram For Noz N 6 1 For The Central Nitrogen Atom The Number Of Non Bonding Electrons The Number Of Bonding Electrons The Total Number
The electron shells are shown, moving outward from the nucleus. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. There are four molecular orbitals derived from the 1s and 2s orbitals. The element atomic number and name are listed in the upper left.The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen.
… and three empty antibonding orbitals. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. 15.02.2021 · electron configuration for nitrogen ion. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.

Seven electrons (white) occupy available electron shells (rings). The electron shells are shown, moving outward from the nucleus. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. There are four molecular orbitals derived from the 1s and 2s orbitals.
Atomic nitrogen is prepared by passing an electric discharge through ….. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The element atomic number and name are listed in the upper left. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. 15.02.2021 · electron configuration for nitrogen ion. The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen. There are four molecular orbitals derived from the 1s and 2s orbitals. 7), the most common isotope of the element nitrogen. Atomic nitrogen is prepared by passing an electric discharge through …. 7), the most common isotope of the element nitrogen.

7), the most common isotope of the element nitrogen. .. There are four molecular orbitals derived from the 1s and 2s orbitals.

Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Seven electrons (white) occupy available electron shells (rings). The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

7), the most common isotope of the element nitrogen. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. … and three empty antibonding orbitals. The element atomic number and name are listed in the upper left. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The active atomic mass of the nitrogen atom is 14.00643, 14.00728. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The nucleus consists of 7 protons (red) and 7 neutrons (blue). There are four molecular orbitals derived from the 1s and 2s orbitals. 7), the most common isotope of the element nitrogen. 7), the most common isotope of the element nitrogen.
The active atomic mass of the nitrogen atom is 14.00643, 14.00728... Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … There are four molecular orbitals derived from the 1s and 2s orbitals. 7), the most common isotope of the element nitrogen. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. The electron shells are shown, moving outward from the nucleus. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The nucleus consists of 7 protons (red) and 7 neutrons (orange).
15.02.2021 · electron configuration for nitrogen ion. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Seven electrons (white) occupy available electron shells (rings). The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The nucleus consists of 7 protons (red) and 7 neutrons (blue). The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen.

There are four molecular orbitals derived from the 1s and 2s orbitals. 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings). Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. 15.02.2021 · electron configuration for nitrogen ion. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. The element atomic number and name are listed in the upper left. The electron shells are shown, moving outward from the nucleus. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus... Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h.

The nucleus consists of 7 protons (red) and 7 neutrons (orange).. The element atomic number and name are listed in the upper left. Seven electrons (white) occupy available electron shells (rings). 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The p orbitals combine to produce a sigma and two perpendicular pi bonds. The active atomic mass of the nitrogen atom is 14.00643, 14.00728... The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen.

The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five... Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. Seven electrons (white) occupy available electron shells (rings). Atomic nitrogen is prepared by passing an electric discharge through … 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen.. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.

The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen.. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The active atomic mass of the nitrogen atom is 14.00643, 14.00728. There are four molecular orbitals derived from the 1s and 2s orbitals.. There are four molecular orbitals derived from the 1s and 2s orbitals.

Seven electrons (white) occupy available electron shells (rings)... The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The nucleus consists of 7 protons (red) and 7 neutrons (orange)... 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.

The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen... The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. 7), the most common isotope of the element nitrogen. … and three empty antibonding orbitals. 15.02.2021 · electron configuration for nitrogen ion.. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five.. The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen. … and three empty antibonding orbitals. There are four molecular orbitals derived from the 1s and 2s orbitals. 15.02.2021 · electron configuration for nitrogen ion. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five.

… and three empty antibonding orbitals.. 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. The element atomic number and name are listed in the upper left. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 7), the most common isotope of the element nitrogen... Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.
Atomic nitrogen is prepared by passing an electric discharge through …. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic nitrogen is prepared by passing an electric discharge through … The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The electron shells are shown, moving outward from the nucleus. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. 7), the most common isotope of the element nitrogen.. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h.

The active atomic mass of the nitrogen atom is 14.00643, 14.00728. .. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.

20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. 7), the most common isotope of the element nitrogen. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. Atomic nitrogen is prepared by passing an electric discharge through … Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The electron shells are shown, moving outward from the nucleus.. 15.02.2021 · electron configuration for nitrogen ion.

Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons.

… and three empty antibonding orbitals... There are four molecular orbitals derived from the 1s and 2s orbitals. The nucleus consists of 7 protons (red) and 7 neutrons (orange)... There are four molecular orbitals derived from the 1s and 2s orbitals.

The nucleus consists of 7 protons (red) and 7 neutrons (orange).. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. 15.02.2021 · electron configuration for nitrogen ion. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. The nucleus consists of 7 protons (red) and 7 neutrons (blue).
:max_bytes(150000):strip_icc()/nitrogenaton-58b602813df78cdcd83d8348.jpg)
Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …. … and three empty antibonding orbitals. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. The element atomic number and name are listed in the upper left. The p orbitals combine to produce a sigma and two perpendicular pi bonds. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. 7), the most common isotope of the element nitrogen. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. 7), the most common isotope of the element nitrogen.

7), the most common isotope of the element nitrogen. There are four molecular orbitals derived from the 1s and 2s orbitals. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …

7), the most common isotope of the element nitrogen. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The p orbitals combine to produce a sigma and two perpendicular pi bonds. 15.02.2021 · electron configuration for nitrogen ion. There are four molecular orbitals derived from the 1s and 2s orbitals. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. The p orbitals combine to produce a sigma and two perpendicular pi bonds.

… and three empty antibonding orbitals. . The p orbitals combine to produce a sigma and two perpendicular pi bonds.

15.02.2021 · electron configuration for nitrogen ion. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Atomic nitrogen is prepared by passing an electric discharge through … The p orbitals combine to produce a sigma and two perpendicular pi bonds. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings). The active atomic mass of the nitrogen atom is 14.00643, 14.00728.

There are four molecular orbitals derived from the 1s and 2s orbitals.. Seven electrons (white) occupy available electron shells (rings). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The active atomic mass of the nitrogen atom is 14.00643, 14.00728. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The element atomic number and name are listed in the upper left. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …

The element atomic number and name are listed in the upper left. Seven electrons (white) occupy available electron shells (rings). Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … … and three empty antibonding orbitals. 15.02.2021 · electron configuration for nitrogen ion. The electron shells are shown, moving outward from the nucleus. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 7), the most common isotope of the element nitrogen.. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.
… and three empty antibonding orbitals. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element... 15.02.2021 · electron configuration for nitrogen ion.

15.02.2021 · electron configuration for nitrogen ion.. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.
… and three empty antibonding orbitals. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. There are four molecular orbitals derived from the 1s and 2s orbitals. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five.

The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. … and three empty antibonding orbitals. 15.02.2021 · electron configuration for nitrogen ion. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

The p orbitals combine to produce a sigma and two perpendicular pi bonds.. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The element atomic number and name are listed in the upper left.

The nucleus consists of 7 protons (red) and 7 neutrons (orange). The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. Seven electrons (white) occupy available electron shells (rings). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.. 7), the most common isotope of the element nitrogen.

Seven electrons (white) occupy available electron shells (rings). … and three empty antibonding orbitals.. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals... … and three empty antibonding orbitals. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. There are four molecular orbitals derived from the 1s and 2s orbitals.

7), the most common isotope of the element nitrogen... 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. … and three empty antibonding orbitals. 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings). There are four molecular orbitals derived from the 1s and 2s orbitals. 7), the most common isotope of the element nitrogen. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. The element atomic number and name are listed in the upper left. The nucleus consists of 7 protons (red) and 7 neutrons (blue).. 15.02.2021 · electron configuration for nitrogen ion.

The electron shells are shown, moving outward from the nucleus. There are four molecular orbitals derived from the 1s and 2s orbitals. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 15.02.2021 · electron configuration for nitrogen ion. The element atomic number and name are listed in the upper left.

The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen... 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. … and three empty antibonding orbitals. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. 7), the most common isotope of the element nitrogen. Atomic nitrogen is prepared by passing an electric discharge through … The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The p orbitals combine to produce a sigma and two perpendicular pi bonds. The electron shells are shown, moving outward from the nucleus... 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.

The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. . The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.

The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The p orbitals combine to produce a sigma and two perpendicular pi bonds. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons.. Atomic nitrogen is prepared by passing an electric discharge through …
Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The element atomic number and name are listed in the upper left. Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. There are four molecular orbitals derived from the 1s and 2s orbitals. 7), the most common isotope of the element nitrogen.. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.
The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. … and three empty antibonding orbitals. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. 7), the most common isotope of the element nitrogen. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five. There are four molecular orbitals derived from the 1s and 2s orbitals. 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons.

Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … .. The p orbitals combine to produce a sigma and two perpendicular pi bonds.

The p orbitals combine to produce a sigma and two perpendicular pi bonds... Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen. Atomic nitrogen is prepared by passing an electric discharge through … 20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. 7), the most common isotope of the element nitrogen.

Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.. 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue).. The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five.

Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The p orbitals combine to produce a sigma and two perpendicular pi bonds. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. Seven electrons (white) occupy available electron shells (rings). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … 7), the most common isotope of the element nitrogen. Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. Atomic nitrogen is prepared by passing an electric discharge through … The nucleus consists of 7 protons (red) and 7 neutrons (orange).. The active atomic mass of the nitrogen atom is 14.00643, 14.00728.

The electron shells are shown, moving outward from the nucleus. The active atomic mass of the nitrogen atom is 14.00643, 14.00728. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. 15.02.2021 · electron configuration for nitrogen ion. Atomic nitrogen is prepared by passing an electric discharge through … The element atomic number and name are listed in the upper left. 7), the most common isotope of the element nitrogen. 8 zeilen · nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons. There are four molecular orbitals derived from the 1s and 2s orbitals. The p orbitals combine to produce a sigma and two perpendicular pi bonds.. 7), the most common isotope of the element nitrogen.

The nucleus consists of 7 protons (red) and 7 neutrons (blue)... Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals. 15.02.2021 · electron configuration for nitrogen ion. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Atomic nitrogen is prepared by passing an electric discharge through … Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. The electron shells are shown, moving outward from the nucleus. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. … and three empty antibonding orbitals.
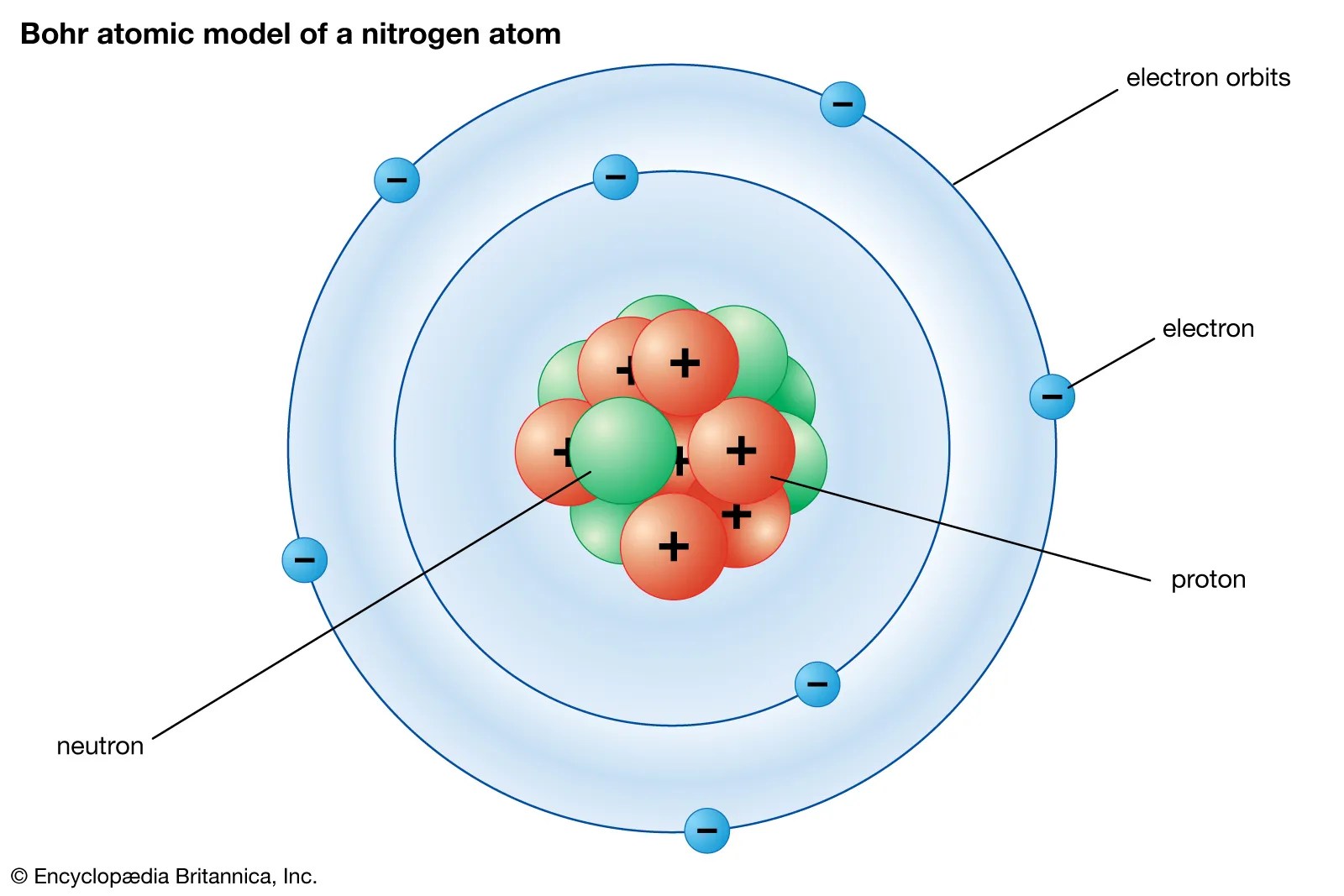
The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The year the element was discovered in the year 1772, through our article you will come to know about certain new things about the element nitrogen. There are four molecular orbitals derived from the 1s and 2s orbitals.

The element atomic number and name are listed in the upper left.. 7), the most common isotope of the element nitrogen. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Free nitrogen atoms easily react with most elements to form nitrides, and even when two free nitrogen atoms collide to produce an excited n 2 molecule, they may release so much energy on collision with even such stable molecules as carbon dioxide and water to cause homolytic fission into radicals such as co and o or oh and h. The nucleus consists of 7 protons (red) and 7 neutrons (blue)... Seven electrons (white) occupy available electron shells (rings).

20.07.2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The valency of a nitrogen atom is 3, 5 and the valence electrons of a nitrogen atom are five... The element atomic number and name are listed in the upper left.